Application note
Rapid-C+: Supporting Annex 1 Compliance Through Continuous Real-Time Viable Air Monitoring
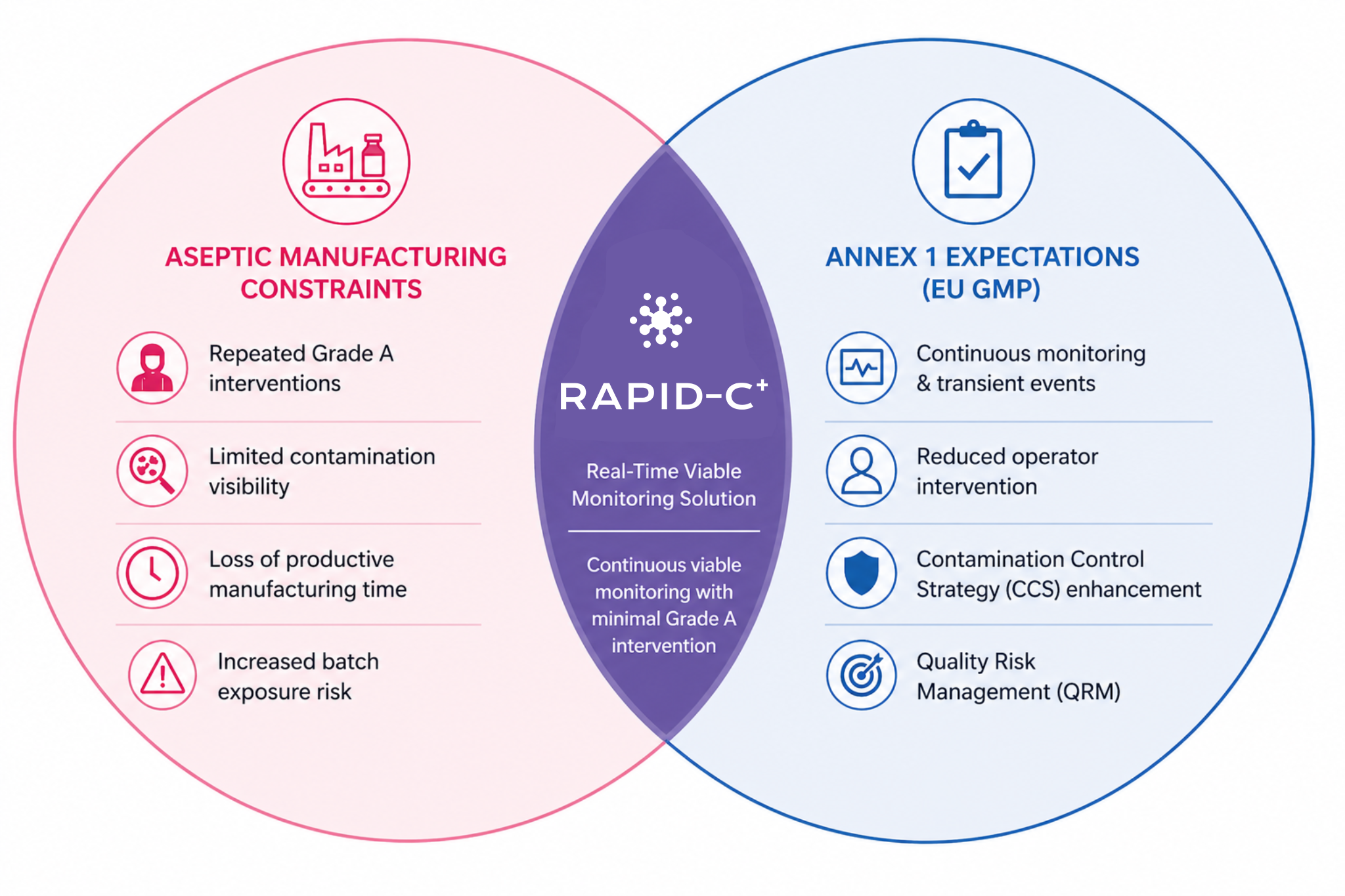
The revised European Union GMP Annex 1: Manufacture of Sterile Medicinal Products [1] became effective on 25 August 2023. It reinforces the expectation for advanced technologies capable of strengthening contamination control strategies (CCSs). Annex 1 encourages the adoption of technologies like Restricted Access Barrier Systems (RABS), isolators (§4.3), robotic systems, rapid/ alternative methods, and continuous monitoring systems to fortify product protection against potential contaminants (§2.1).
Within this framework, real-time monitoring provides pharmaceutical manufacturers with a proactive means of identifying, scientifically evaluating, and mitigating potential risks to the quality of the product as part of Quality Risk Management (QRM) (§2.2). This is of special importance for high-value or short shelf life products, where traditional environmental monitoring data may not be readily available (§10.10).
Rapid-C+ is designed to support this Annex 1 expectation by operating in real time and enabling continuous viable air monitoring in Grade A for the entire duration of critical processing operations, including aseptic set-up and assembly (§9.24).
Discover the full application note by following the link below.
